Related Resources: fluid flow
Capillary Action Review and Equations
Capillary Action is due to both the cohesive forces between liquid molecules and adhesive forces of liquid molecules. It shows up as the difference in liquid surface elevations between the inside and outside of a small tube that has one end submerged in the liquid (Fig. 1).
Capillarity is commonly expressed as the height of this rise. In equation form,
h = ( 2 · σ · cosθ ) / [ ( w1 - w2 ) r ]
Where:
h = capilary rise, ft (m),
σ = surface tension, lb/ft (N/m),
w1 and w2 = specific weights of fluids below and above meniscus, respectively, ft (m),
θ = angle of contact,
r = radius of capillary tube, ft (m)
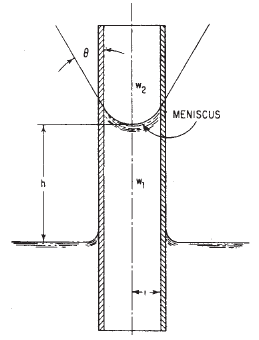
Figure 1
Capillary action raises water in a small-diameter tube. Meniscus, or liquid surface, is concave upward.
Reference:
Civil Engineering Formulas SEcond Edition, Tyler G. Hicks, P.E.